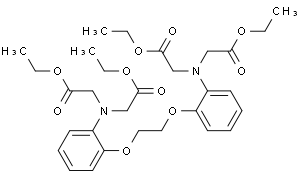
BAPTA tetraethyl ester
BAPTA tetraethyl ester
CAS: 73630-07-6
Molecular Formula: C30H40N2O10
BAPTA tetraethyl ester - Names and Identifiers
| Name | BAPTA tetraethyl ester |
| Synonyms | CS-964 NSC 368732 BAPTA TETRAETHYL ESTER BAPTA tetraethyl ester 1,2-bis(2-aminophenoxy)ethane-n,n,n',n'-tetraacetic acid tetraethyl ester tetraethyl 2,2',2'',2'''-[ethane-1,2-diylbis(oxybenzene-2,1-diylnitrilo)]tetraacetate N,N'-[1,2-Ethanediylbis(oxy-2,1-phenylene)]bis[N-(2-ethoxy-2-oxoethyl)glycine diethyl ester Tetraethyl 2,2',2'',2'''-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))bis(azanetriyl))tetraacetate NSC 368732 N,N'-[1,2-Ethanediylbis(oxy-2,1-phenylene)]bis[N-(2-ethoxy-2-oxoethyl)glycine diethyl ester |
| CAS | 73630-07-6 |
| InChI | InChI=1/C30H40N2O10/c1-5-37-27(33)19-31(20-28(34)38-6-2)23-13-9-11-15-25(23)41-17-18-42-26-16-12-10-14-24(26)32(21-29(35)39-7-3)22-30(36)40-8-4/h9-16H,5-8,17-22H2,1-4H3 |
BAPTA tetraethyl ester - Physico-chemical Properties
| Molecular Formula | C30H40N2O10 |
| Molar Mass | 588.65 |
| Density | 1.21g/cm3 |
| Melting Point | 98-100°C |
| Boling Point | 657.4°C at 760 mmHg |
| Flash Point | 351.4°C |
| Vapor Presure | 3.69E-17mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.55 |
| MDL | MFCD00532663 |
BAPTA tetraethyl ester - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
BAPTA tetraethyl ester - Introduction
BAPTA tetraethyl ester(BAPTA tetraethyl ester) is an organic compound that is a chelating agent. The following is a description of its nature, use, preparation and safety information:
Nature:
-Chemical formula: C40H58N2O14
-Molecular weight: 826.9g/mol
-Appearance: White solid
Use:
BAPTA tetraethyl ester is mainly used for the chelation and research of calcium ions in cells and biological materials. It can pass through the cell membrane permeability is good, into the cell, and can selectively react with calcium ions to form a stable chelate, thereby controlling the concentration of intracellular calcium ions. It is widely used in the field of biomedical research, especially in the study of calcium signaling pathways, apoptosis and cell proliferation.
Preparation Method:
The preparation of BAPTA tetraethyl ester usually involves a multi-step reaction. The main steps include the Archimedes reaction of benzaldehyde to prepare dione compounds, and then the amination reaction to generate dione amines. Finally, BAPTA tetraethyl ester was obtained by reaction with tetraacetic anhydride.
Safety Information:
BAPTA tetraethyl ester is a chemical and should be operated under suitable laboratory conditions. It is necessary to take appropriate protective measures during use, such as wearing protective glasses and gloves. In addition, its specific toxicity and hazard information may vary from supplier to supplier, so it is necessary to read the product manual carefully and follow the correct operation and disposal regulations. For safety reasons, users should keep it in a safe place and avoid contact with incompatible substances. In case of any discomfort or accident, seek professional help immediately.
Nature:
-Chemical formula: C40H58N2O14
-Molecular weight: 826.9g/mol
-Appearance: White solid
Use:
BAPTA tetraethyl ester is mainly used for the chelation and research of calcium ions in cells and biological materials. It can pass through the cell membrane permeability is good, into the cell, and can selectively react with calcium ions to form a stable chelate, thereby controlling the concentration of intracellular calcium ions. It is widely used in the field of biomedical research, especially in the study of calcium signaling pathways, apoptosis and cell proliferation.
Preparation Method:
The preparation of BAPTA tetraethyl ester usually involves a multi-step reaction. The main steps include the Archimedes reaction of benzaldehyde to prepare dione compounds, and then the amination reaction to generate dione amines. Finally, BAPTA tetraethyl ester was obtained by reaction with tetraacetic anhydride.
Safety Information:
BAPTA tetraethyl ester is a chemical and should be operated under suitable laboratory conditions. It is necessary to take appropriate protective measures during use, such as wearing protective glasses and gloves. In addition, its specific toxicity and hazard information may vary from supplier to supplier, so it is necessary to read the product manual carefully and follow the correct operation and disposal regulations. For safety reasons, users should keep it in a safe place and avoid contact with incompatible substances. In case of any discomfort or accident, seek professional help immediately.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: BAPTA tetraethyl ester Visit Supplier Webpage Request for quotationCAS: 73630-07-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,2-Bis(2-pyridyl)ethylene Request for quotation
CAS: 73630-07-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 73630-07-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BAPTA TETRAETHYL ESTER Request for quotation
CAS: 73630-07-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 73630-07-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Bapta Tetraethyl Ester Visit Supplier Webpage Request for quotation
CAS: 73630-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 73630-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: BAPTA tetraethyl ester Visit Supplier Webpage Request for quotationCAS: 73630-07-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,2-Bis(2-pyridyl)ethylene Request for quotation
CAS: 73630-07-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 73630-07-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BAPTA TETRAETHYL ESTER Request for quotation
CAS: 73630-07-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 73630-07-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Bapta Tetraethyl Ester Visit Supplier Webpage Request for quotation
CAS: 73630-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 73630-07-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



